(E12) Electricity and Chemistry
On the scale of atoms and molecules, electric forces rule the behavior of matter.
Thus, as may be expected, chemistry is very relevant to the study of electricity. This is not a course on chemistry, and will therefore assume the user has previous acquaintance with fundamental chemical facts, without going into many details. One such fact is that matter consists of tiny atoms, which come in 91 stable kinds or "elements" (not 92, since Technetium is unstable), plus some unstable ones. Each kind of atoms has its distinct chemical behavior. Some materials consist of a single variety of atoms, but many more are built of molecules, each combining several (different or identical) atoms ("chemical compounds"). The type of atom or molecule determines the properties of the material, which can also depend on temperature and other factors. Each element has a name, and chemical formulas label their atoms by one or two letters (first one always a capital letter). For example:
O for Oxygen N for Nitrogen C for Carbon He for Helium Ar for Argon Fe for iron (Ferrum in Latin) Na for sodium (Natrium) Cl for Chlorine, K for Potassium (Kalium) P for phosphorus Molecules are denoted by combinations of letters, identifying the atoms which make them up. If the molecule contains more than a single atom of certain kind, a subscript on the appropriate formula tells how many: Examples: NaCl for table salt ("sodium chloride") H2O for water (no one calls it dihydrogen oxide--though one could) CO2 carbon dioxide (a gas produced by burning and by breathing) HCl Hydrochloric accid H2SO4 Sulfuric acid (S represents the sulfur atom) NaOH Sodium hydroxide also known as lye. Lye and fats combine to soap. Ca(OH)2 Calcium hydroxide, from quicklime and water-- quicklime being the powder left after roasting limestone in a furnace ("kiln"). Important in mortar and building materials. CaSO4 Gypsum, used in walls ("drywall") N2 Nitrogen, a gas whose atoms combine in pairs. O2 Oxygen, another such gas. Air is about 78% nitrogen, 21% oxygen, and most of the rest is argon Ar, whose atoms don't combine. H2 Hydrogen, the lightest gas of all. CH4 Methane, which burns and is found in natural gas of oil wells.
It should be realized that the above statements distill the work of many generations. We also won't go into the history of those discoveries, because the goal now is to explain how electric cells and batteries were discovered, and how electricity is harnessed by chemical processes
It also dissolves a wide class of compounds. Mixing different water solutions (sometimes adding heat--as in the cooking of food) was one easy way of creating new compounds, and three groups of solutions turned out to be good conductors of electricity--acids, bases (or alkalis) and salts. Since the early 1900s much more is known about matter, especially that (discovery by Rutherford, 1911) each atom consists of a compact positively charged nucleus, surrounded by negative electrons. The electrons are relatively lightweight, and most of the mass or weight of matter is in the nuclei of its atoms. They are held together by electrical attraction between (+) and (–) charges, a situation which has sometimes been compared to the way planets are held by the gravity of the Sun. This analogy is not really accurate, because on the atomic scale, certain new properties ("quantum effects") begin dominating the laws of physics. The location of electrons in atoms is not predictable: all we have is the likelihood of an electron being found at various points, given by a wave function defining (for atoms at rest) certain standing waves. The atoms are stable only if the wave function takes one of a number of symmetric patterns, each with its energy. When atoms combine to molecules, such patterns also exist, and when atoms or molecules combine to form solids, such laws often determine crystal structure, electric conductivity and other properties. The lightest atom is hydrogen, whose positive nucleus is a distinct particle, the proton, 1840 times heavier than the electron. Heavier atoms seem to contain many protons, surrounded by the same number of electrons, so that electrically, the atom (usually) is neither positive nor negative. Actually, the weight of atoms (atomic weight, in units of the proton) is generally double the weight of its protons, or more than double. It turned out (1932) that nuclei also contains neutrons, particles somewhat similar to the protons but with no electric charges, in numbers equal to that of protons, or in heavy atoms, in slightly larger numbers. Again, this has been the work of many generations. Over the 1800s, scientists generated electricity by chemical means, separated compounds in solutions by electrical means and formulated laws governing such process, but did not fully understand their reasons After all, the electron was only discovered in 1896! (the next few paragraphs follow parts of "Positive Ions--History") Ionic CompoundsThe unique chemistry associated with water was explained in 1884 by Svante Arrhenius (1859-1927), a many-talented Swede who received the 1903 Nobel prize for chemistry and who (among his many achievements) first suggested the "greenhouse effect". Arrhenius proposed that when a compound such as table salt NaCl (sodium chloride) was dissolved in water, it broke up into electrically charged "ions" (Greek for "the ones that move") Na+ and Cl-. Electric forces made Na+ ions move in one direction, Cl- ions in the opposite one, and that was how an electric current could be carried. 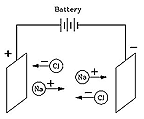
Although at first this seemed like a strange idea, today it is quite well understood. Many chemical molecules are formed when atoms share electrons, but molecules such as those of NaCl are different. There, the sodium atom (Na) gives up an electron to the chlorine (Cl), creating ions Na+ and Cl-, which in solid salt are held together by their electric attraction ("ionic bond"). Water, however, greatly weakens electric forces (on the atomic scale), allowing some ions to drift free whenever salt is dissolved in water, and allowing the water to conduct electricity.
The smallest atomic positive ion is the proton, the nucleus of hydrogen. Substances which when dissolved in water produce ions of hydrogen are known as acids and any such solution (e.g. HCl, H2SO4 ) has a sour taste. Of course, the fraction of acid molecules which actually breaks up into ions in a water solution can vary--it is large in "strong" acids and small in "weak" acids. Even in pure water a tiny fraction of the molecules is split up into ions H+ and (OH)– ("hydroxyl") at any time. The degree of "sourness" depends on the concentration of the acid in the water and on its strength; carbonated water (for instance) has a slightly sour taste, because some CO2 is dissolved in it, creating the weak "carbonic acid" H2CO3.
In addition, many other ionic compounds exist, dissolving in water at least partially. Metals are attacked by acids, replacing their hydrogen atoms with metal ones--e.g. copper and sulfuric acid give CuSO4 (green-blue crystals), and sulfuric acid from burning fuel creates "acid rain", which turns the surface of marble to gypsum and thus damages buildings and outdoor art. Any solution of an ionic compound contains equal charges of positive or negative ions, and therefore carries no net electric charge, but still can conduct an electric current by moving ions across the solution. As ions arrived at the other electrode, they sometimes are deposited (for instance, "electroplating" it with silver or copper) and in general further chemical reactions may occur. It is worth noting that water can also dissolve some non-ionic substances whose atoms contain weak bonds-- sugar or alcohol, for instance. And of course, many compounds are formed by sharing of electrons in a molecule ("covalent bond") and not by the ionic bond, and these (e.g. compounds in glass or in plastics) are usually not dissolved in water.
|